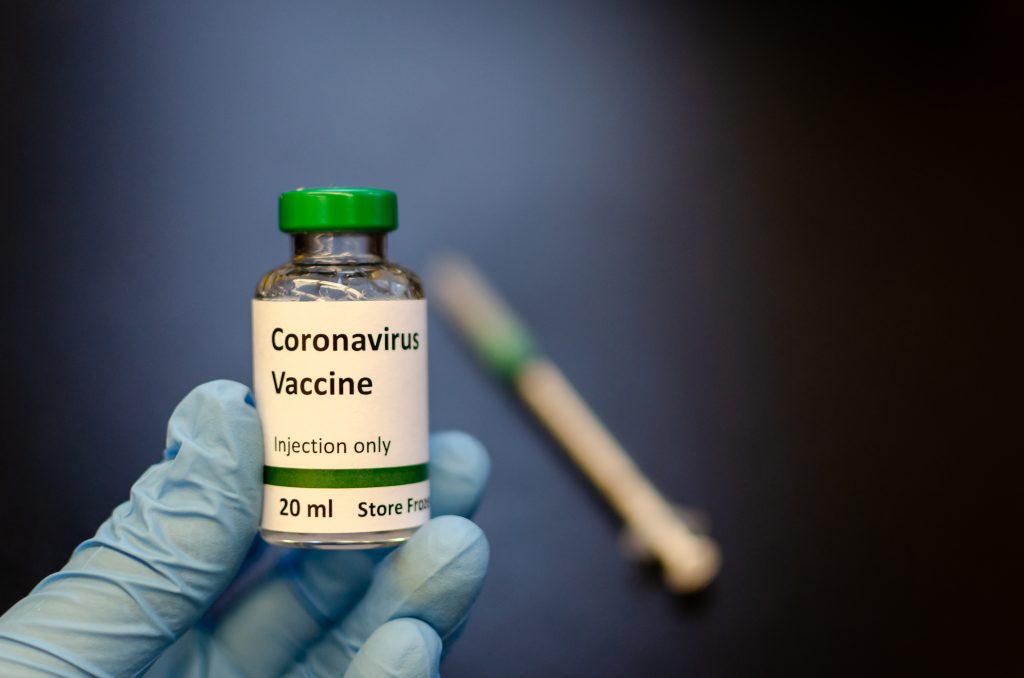
China’s military has gained the green light to apply a COVID-19 vaccine competitor produced by its research unit and CanSino Biologics (6185.HK) after clinical trials confirmed it was safe and revealed some efficacy, the company said on Monday.
The Ad5-nCoV is one of China’s eight vaccine candidates allowed for human trials at home and abroad for the respiratory disease prompted by the new coronavirus. The shot also won approval for human testing in Canada, Reuturs reported.
“The Ad5-nCoV is currently limited to military use only and its use cannot be expanded to a broader vaccination range without the approval of the Logistics Support Department,” CanSino said, referring to the Central Military Commission department which approved the military use of the vaccine.
CanSino declined to disclose whether the injection of the vaccine candidate is compulsory or optional, indicating commercial secrets, in an email to Reuters.
What's happening in Tunisia?
Subscribe to our Youtube channel for updates.